Introduction
Exposure to risk factors during childhood may contribute to the development of future cardiovascular (CV) disease. Thus, obstructive sleep apnea (OSA) could act as a precipitating factor and be a modifiable risk factor. The use of new imaging tools, such as echocardiography, or the incorporation of new biological signals extracted from sleep studies or biomarkers related to inflammation and oxidative stress, may help establish a new approach to the prognosis and treatment of children with OSA. In the era of personalized medicine, identifying subjects at the most significant risk of developing future CV disease can help inform therapeutic decisions aimed at preventing this disease by suppressing this modifiable risk factor or avoiding harm to those less vulnerable. This approach is crucial given that this is a particularly at risk developing population. The objective of this review is to provide an overview of the current evidence on pediatric OSA from a CV perspective and of new tools that, when applied in routine clinical practice, can aid decision-making.
Literature search methodology
An initial MEDLINE search was performed using the keywords “obstructive sleep apnea,” “cardiovascular risk,” “pediatric” and “precision medicine” for the publication years 2000 to 2025. A second MEDLINE search used additional keywords linked to each paragraph and subparagraph of this review, including “phenotypes”, “clinical features”, “diagnosis”, “guidelines”, “treatment”, “po lysomnography”, “biomarkers”, “biosignals”, “prevention”, “inflammation”, “endothelial dysfunction”, “hypertension”, “autonomic dysfunction”, “risk factors”, “comorbidity”, “mortality”, “cardiovascular disease”, “positive airway pressure”, “otolaryngological surgery”, and “obesity”. Data from systematic reviews were highlighted.
Definition, prevalence and etiology of OSA in children
OSA is a type of sleep-disordered breathing (SDB) that is marked by frequent arousals, apneas, and hypopneas1. It can lead to reduced blood oxygen saturation and hypoventilation during sleep in children. The causes, clinical presentation, and potential consequences of OSA in children differ significantly from those in adults. Therefore, it is essential to have a specific approach tailored for this population. As well, although OSA is a common condition in children, with a prevalence ranging from 1% to 4%, it is underdiagnosed2.
The definition of OSA in children varies across available guidelines and has not been updated recently. The severity of OSA in children is typically classified using the apnea-hypopnea index (AHI), which measures the number of respiratory events per hour of sleep on sleep studies. Generally, an AHI equal or more than 1 is considered the normal cutoff for diagnosing OSA. The classification of OSA severity is as follows: mild OSA if the AHI is < 5 events/h, moderate OSA if the AHI is between 5 and 10 events per hour, and severe OSA if the AHI exceeds 10 events/h1–4. The etiology of OSA in children is multifactorial, involving multiple risk factors that contribute to upper airway narrowing and collapsibility. These factors include both anatomical and neuromuscular disturbances, which contribute to increased airway resistance and hinder the normal function of the dilator muscles4. The most common risk factor for pediatric OSA is adenotonsillar hypertrophy, which typically peaks between 2 and 8 years of age, coinciding with the onset of OSA5. Craniofacial abnormalities can also contribute to upper airway narrowing, often present in pediatric syndromes. Obesity has been identified as a significant contributor to OSA, and its prevalence is steadily rising, currently affecting 5.6% of females and 7.8% of males6.
Focusing on the CV sphere of sleep apnea
Pathophysiological bases and preventive medicine
Globally, CV disease is the leading cause of death in adults7. There is evidence that the underlying cause of this disease, atherosclerosis, may begin in childhood8,9. Changes in the autonomic nervous system, vasomotor tone, systemic inflammation, and atherogenesis associated with OSA can lead to endothelial dysfunction. In recent years, evidence has emerged regarding OSA as a possible risk factor10,11, although it has not yet been given the importance it deserves in this regard.
The mechanisms underlying CV issues related to OSA are not yet fully understood, but several potential intermediate mechanisms have been proposed12. Intermittent hypoxia leads to the production of reactive oxygen species and promotes oxidative stress. Additionally, the combined effects of hypoxia and hypercapnia associated with OSA increase sympathetic nervous system activity. This increase is especially pronounced during nighttime apnea episodes, although a rise in daytime sympathetic tone can also be observed. Patients with OSA often experience a pro-inflammatory state, which can be attributed to local inflammatory changes caused by soft-tissue vibrations during snoring. Furthermore, the activation of the sympathetic nervous system contributes to a systemic pro-inflammatory state by stimulating the release of inflammatory mediators. Moreover, hypoxemia, repeated awakenings during sleep, and sympathetic activation lead to the development and persistence of procoagulant inflammatory mediators, further contributing to a procoagulant state. Finally, it is important to note that hypoxemia, sympathetic activation, and disrupted sleep contribute to endothelial dysfunction and metabolic issues, increasing the risk of CV complications. These mechanisms are responsible for a pathological CV response12,13 and, in the pediatric population, can trigger cardiological alterations at the structural, hemodynamic and functional levels, increased blood pressure (BP), especially during the night, and endothelial dysfunction14. Based on this idea, the treatment of OSA could reduce the activation of these intermediate pathways and, consequently, the development of CV disease.
CV comorbidities and consequences of OSA in children
OSA in children is linked to various adverse health outcomes, including behavioral and neurocognitive disorders, growth retardation, CV diseases, and metabolic issues. These conditions, collectively, diminish the child’s quality of life and increase future CV risk15. The strongest evidence available relates to changes in BP, heart rhythm disturbances, and metabolic syndrome.
Children with OSA often display elevated systolic and diastolic BP, increased BP variability, and diminished BP dipping during sleep14. This is because recurrent episodes of hypoxemia and hypercapnia in children with such conditions activate the sympathetic nervous system, leading to increased levels of catecholamines16. In children with OSA, we observe higher BP values both during the day and at night. Specifically, there are increased systolic BP levels during sleep and a reduced drop in systolic and mean BP at night14,17,18. Elevated BP levels during the day and at night contribute to the development of hypertension Therefore, monitoring the BP of children with OSA is crucial for identifying those at risk of developing clinically significant elevated BP in adulthood.
Cardiac arrhythmias can occur in children with OSA, but their actual frequency and clinical importance are not well established. Research suggests that most arrhythmias in pediatric OSA are mild and benign, such as sinus arrhythmia or sinus bradycardia. In contrast, more severe OSA may be associated with greater heart rate variability and subtle electrical changes18–20. Increased QT dispersion has been linked to a higher risk of ventricular arrhythmias and has been observed in children with severe OSA. Similarly, greater P-wave dispersion has been reported in a study involving children with severe OSA. These findings suggest that cardiac arrhythmias in pediatric OSA exist along a spectrum – typically mild and benign, but potentially more significant in severe cases19,21. Further studies are needed to understand how OSA contributes to cardiac rhythm disturbances in children.
Finally, children with OSA are at a significantly higher risk of developing early metabolic syndrome, with this risk being six times greater than that of healthy adolescents22,23.
To reduce the harmful consequences of OSA, it is essential to ensure early and accurate diagnosis and effective treatment. The most substantial evidence indicates that this condition can have a considerable impact on CV health during childhood and into adulthood24. Therefore, tools for identifying at-risk children and guidelines for preventative treatment are necessary.
Diagnosis of OSA: implications of the CV sphere
Clinical manifestations
Figure 1 shows the daytime and nighttime symptoms most commonly observed in children with OSA4. At night, they often experience snoring (the most common), witnessed apneas, gasping, mouth breathing, restless sleep, nightmares, and enuresis, which usually improve with treatment. During the day, OSA is linked to behavioral problems such as irritability, aggression, hyperactivity, and depression, along with difficulties in attention, learning, mood regulation, and fatigue. The use of validated parent-completed questionnaires can be useful for systematically collecting symptoms. Physical examination can provide additional information. As we have previously mentioned, alterations in BP, especially the non-dipper pattern, can be important clues to suspect OSA. Obesity and morphological alterations of the oral cavity and thorax, whether or not in the context of syndromic conditions, can also raise our level of suspicion and guide us toward management.
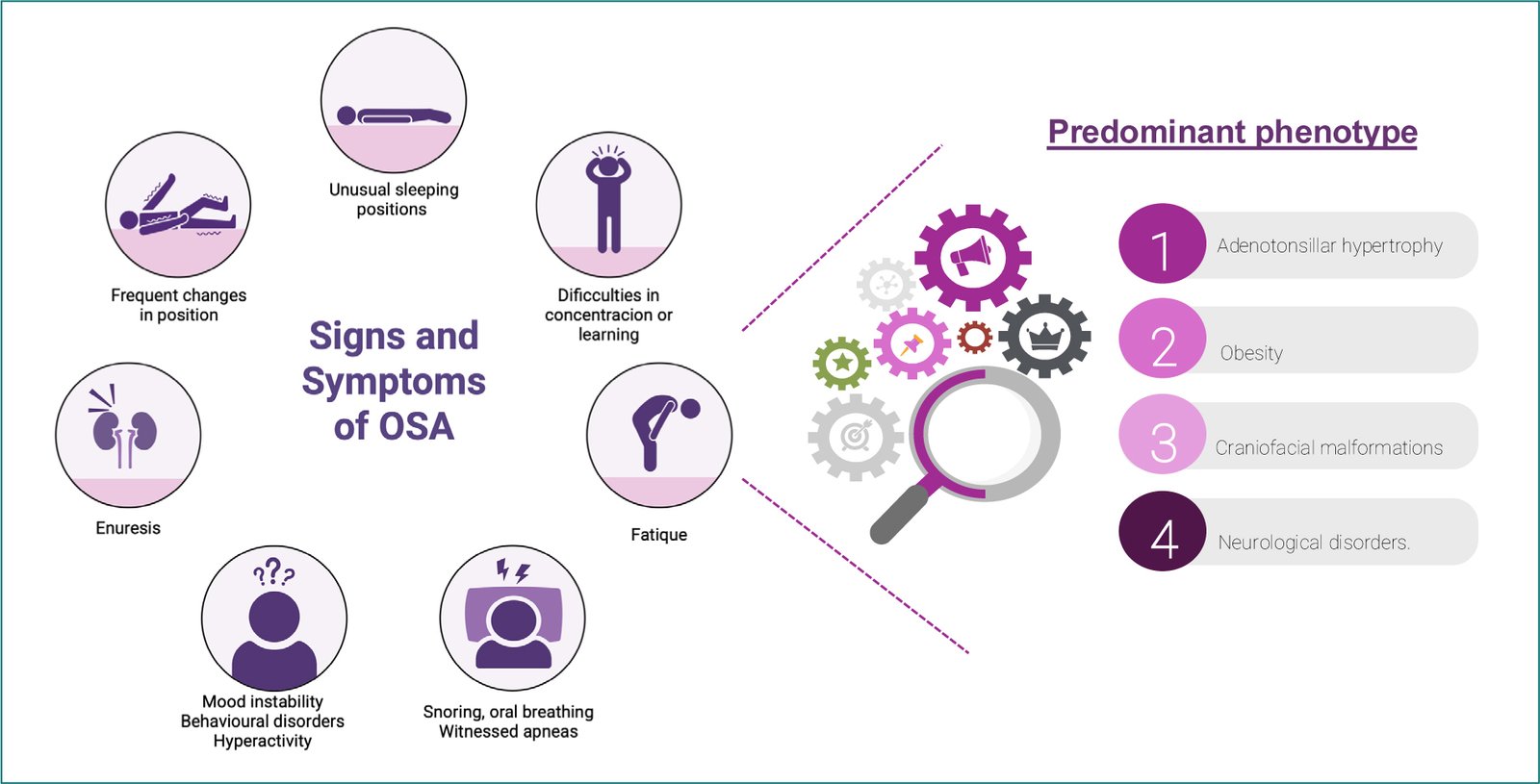
Figure 1. Main symptoms of OSA in children and clinical phenotypes. OSA: obstructive sleep apnea.
Traditional diagnostic methodology
In terms of diagnostic management, polysomnography (PSG) remains the gold standard test. However, due to the limited availability of accredited sleep laboratories for children and the considerable resources required to perform PSG, significant efforts have been made to develop alternative diagnostic methods that are more accessible. While respiratory poligraphy tests have been created, they have not been sufficiently evaluated for general use in the pediatric population4,25. They have been used in Europe as a valid alternative to PSG, while the American Academy of Sleep Medicine maintains that these tests may underdiagnose OSA in children. Other modalities, such as nocturnal oximetry recordings, present a promising option due to their low cost and ease of use. Various studies have explored this approach; however, further validation of the results is necessary26,27.
New tools available to identify subjects at higher CV risk
BP AND BIOSIGNALS: HYPOXIC BURDEN & HEART RATE VARIABILITY
In a secondary analysis of the Kids Trial clinical trial, it has been shown that there exists an association between BP levels and the AHI28. These results revealed that OSA severity was related to a higher risk of NDP.
In recent years, the AHI, as a CV risk marker, has been questioned because it does not measure the immediate consequences of each respiratory event, such as intermittent hypoxemia, or the future CV consequences. Therefore, new markers have emerged that help overcome these limitations, and hypoxic burden (HB) could be proposed as a good marker of future CV risk in children with OSA. HB is obtained from the sleep study by summing individual desaturation areas and dividing by total sleep time, with units of HB being %min/h29. This parameter quantifies the cumulative impact of OSA-related hypoxemia by incorporating the frequency, duration, and depth of oxygen desaturation events. In the adult population over the age of 18, there is solid scientific evidence of its role as a predictor of CV disease and death; however, it is only just beginning to be explored in children29,30.
More recently, in the same cohort of the Kids Trial Study, HB values in pediatric patients with OSA were reported for the first time, and several novel aspects were highlighted31. On the one hand, higher levels of HB were associated with alterations in nocturnal BP dipping, a recognized marker of CV risk. As expected, because desaturations in children are less severe than in adults, HB values in children are lower than in adults. In that cohort, HB greater than 22.53 %min/h was associated with a smaller nocturnal decrease in global mean BP, emphasizing the clinical relevance of HB in estimating CV outcomes in children with OSA and in detecting BP alterations. In addition, HB determination is simple through automated analysis, reproducible, and applicable. It is evident that there is a need to continue researching this parameter in the pediatric population as a marker of future CV risk, but these findings highlight the importance of new research that could simplify clinical practice for these patients, better identify subjects at risk, and guide management cost-effectively.
As we previously mentioned, the frequency and significance of arrhythmias in pediatric OSA remain unclear. Most evidence indicates these arrhythmias are usually mild, such as sinus arrhythmia or bradycardia. However, severe OSA may be associated with increased heart rate variability and subtle electrical changes, such as prolonged QT and P-wave dispersion, which are linked to a higher risk of significant arrhythmias. Overall, although most cardiac arrhythmias in pediatric OSA are benign, more research is needed to fully understand their implications.
SERUM AND URINE BIOMARKERS
In recent years, attempts have been made to understand the pathophysiological mechanisms of OSA in the development of endothelial and endocrine dysfunction through the study of biomarkers18. Some easily accessible serum or urinary inflammatory biomarkers in clinical practice could play a role in CV risk stratification in children with OSA.
Microalbuminuria refers to a mild level of urinary protein loss. A pediatric study compared children with OSA to those without it (the control group) and found that children with moderate to severe OSA are at a higher risk of developing microalbuminuria32. The higher risk may be linked to changes in glomerular permeability, potentially due to hypoxemia during sleep, as well as to sympathetic nervous system activation caused by OSA29. In a meta-analysis of OSA and kidney outcomes in adults, a consistent association was observed between OSA and increased levels of albuminuria or proteinuria, along with a decrease in glomerular filtration rate33.
Untreated OSA increases the risk of insulin resistance and alterations in the regulation of the hypothalamic-pituitary-adrenal axis18. Efforts have been made to understand the bidirectional mechanisms between obesity and OSA that promote these alterations in the endocrine-metabolic system. The complex relationship between OSA, obesity, hypertension, dyslipidemia, and insulin resistance can contribute to metabolic syndrome, leading to elevated serum insulin levels, increased BP, higher triglycerides, and lower high-density lipoprotein levels21. The development of metabolic syndrome associated with OSA can be attributed to several mechanisms, including increased sympathetic activity, elevated serum cortisol levels due to hypothalamic-pituitary-adrenocortical axis activation, and the formation of reactive oxygen species34. Obesity is likely the primary factor linking OSA and metabolic syndrome35. C-reactive protein (CRP), body fat (adiposity), leptin levels, and insulin resistance have been associated with SDB, even after accounting for various demographic factors36,37. CRP is one of the most extensively researched inflammatory markers, and levels of this marker increase with OSA, and CRP may decrease after appropriate treatment38.
ECHOCARDIOGRAPHY
In a systematic review, Castillo-García et al.14 assessed the CV risk of children with OSA using echocardiographic morphological or functional changes. OSA can produce morphological alterations in both ventricles that may worsen with the severity of OSA and improve with treatment. Research by Amin et al.,39,40 found that left ventricular (LV) mass increases with the presence of OSA in proportion to the severity of OSA measured by AHI. These changes were reversible after treatment. In this regard, Chat et al.39 found a relative increase in wall thickness and the interventricular septal index in children with moderate-severe OSA (AHI > 5/h) compared with mild OSA (AHI 1-5/h), which improved after treatment. Domany et al. in 202141 reported similar findings, showing an increase in ventricular mass with increasing OSA severity. Concerning LV function, studies by Amin et al.39,41 identified a decrease in LV diastolic function. They found that the E/A ratio, which measures flow velocity across the mitral valve, was reduced in children with primary snoring (AHI < 1/h) and in those with OSA (AHI ≥ 1/h). Notably, this dysfunction improved only in the OSA group after treatment, while children with primary snoring did not experience similar improvements. Similarly, Ugur et al.42 reported diastolic dysfunction in children with OSA (AHI > 2/h), evidenced by an increase in the mitral E/A ratio, with improvements after treatment. Chan et al.43 also documented alterations in diastolic function. In a study by Kaditis et al.44, decreased LV systolic function was observed in moderate-to-severe OSA (AHI > 5/h) compared with primary snoring (AHI < 2/h). This was measured using lower LV ejection fraction, which represents the volume ejected during systole in relation to the volume in the ventricle at the end of diastole, and LV shortening fraction, indicating LV contractility. In addition, Villa et al.45 demonstrated LV dysfunction in children with OSA (AHI ≥ 1/h) compared with those with primary snoring (AHI < 1/h), with decreased E/A ratios and increased isovolumetric relaxation times. More recently, Domany et al.41 reported that in children with OSA (AHI > 1/h), LV diastolic dysfunction worsened with OSA severity, but improvements were noted after treatment, as indicated by a decrease in the E/e’ ratio during follow-up.
While many studies have reported positive results regarding the CV impacts of outcomes, not all findings have been consistent. Some researchers did not find a correlation between abnormalities detected in preoperative echocardiography and the severity of OSA, nor a relationship between surgical intervention and cardiac impairments related to SDB42,46,47.
In summary, OSA has been shown to trigger cardiac alterations affecting both the LV and right ventricle. These alterations include impairments in diastolic and systolic functions, increased mass. The severity of these disturbances typically worsens with increasing OSA severity, as measured by the AHI. Many studies have evaluated the impact of treatment, noting overall improvement in children when properly managed. Thus, OSA appears to be a significant risk factor for CV health, and treatment for OSA may benefit heart health in this population.
Management of OSA
The approach to treating a child with OSA should focus on identifying the primary phenotype and CV risk factors associated with the condition. In addition, it is crucial to consider factors that may help predict treatment outcomes and the likelihood of OSA persisting.
For children with adenotonsillar hypertrophy, adenotonsillectomy is the first-line treatment4 and improves the AHI to ≤ 1 in approximately 50-70% of cases. However, the procedure’s effectiveness diminishes in younger children, those with severe OSA, chronic asthma, or obesity. In addition, residual disease persists in 20-75% of patients, many of whom continue to snore1,48,49.
Weight management is crucial since weight loss can significantly improve OSA in overweight or obese children and may even be considered a first-line therapy for this demographic50.
Continuous positive airway pressure (CPAP) and bi-level positive airway pressure (BiPAP) can be effective51 in children with craniofacial or neuromuscular disorders or when surgery is not an option or the result is not optimal. CPAP is the most commonly used1,3, while BiPAP is reserved for patients who cannot tolerate CPAP or those experiencing nocturnal hypoventilation2–4,52.
Positional therapy has been widely studied in adults but has limited evidence of effectiveness in children. Nevertheless, it may benefit obese patients or those without tonsillar hypertrophy or with residual OSA53,54.
Orthodontic treatments like rapid maxillary expansion can also help by widening the palate and enhancing airway patency5,55,56.
Medical therapies, including intranasal corticosteroids and montelukast, have demonstrated limited or short-term benefits, especially in non-obese, untreated children, although their clinical relevance remains uncertain57,58. Figure 2 summarizes the available treatment options.
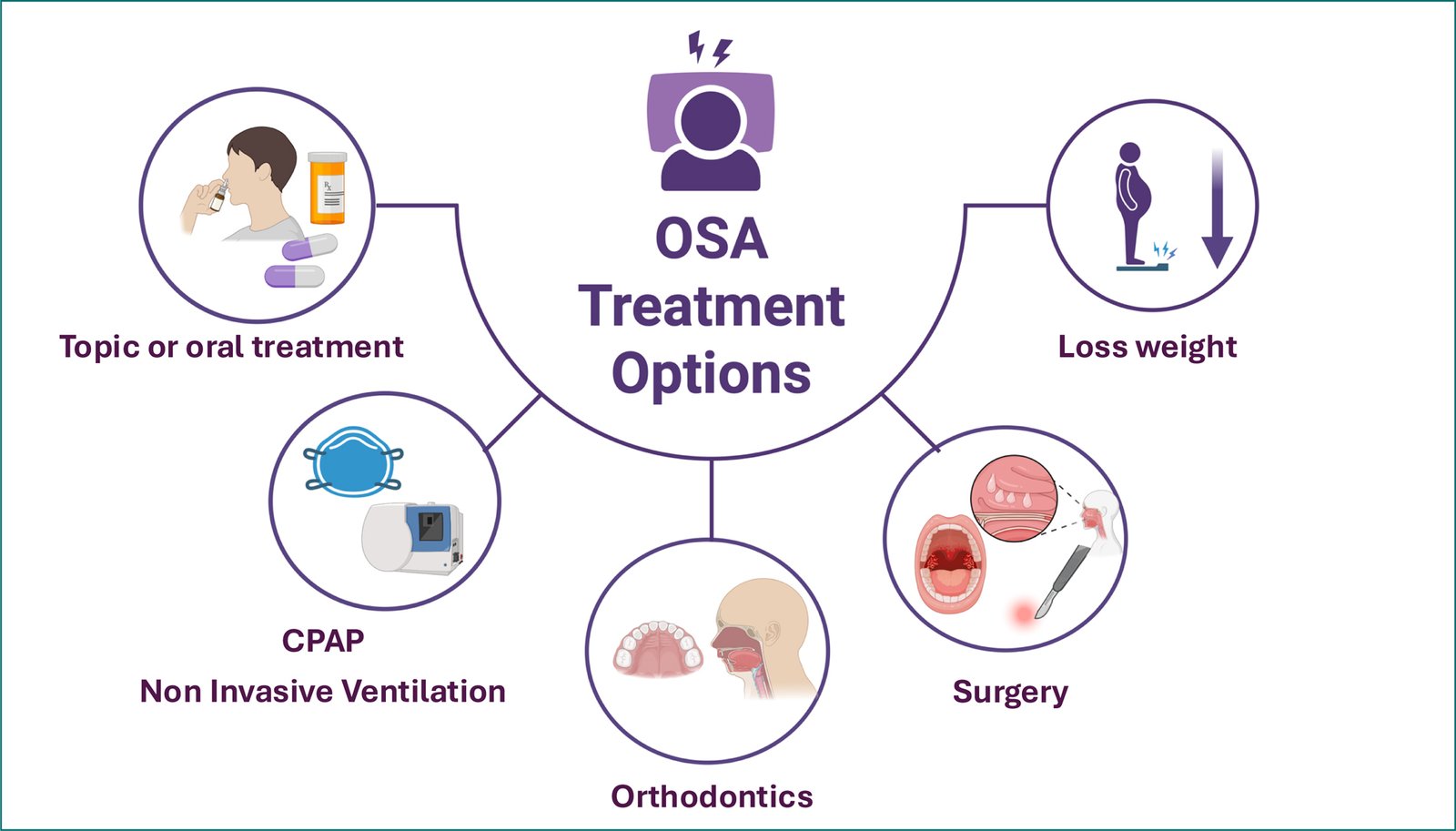
Figure 2. OSA treatment options. CPAP: continous positive airway positive pressure; OSA: obstructive sleep apnea.
The development of updated, evidence-based protocols and new evaluation metrics is essential to optimize diagnosis and treatment strategies, ensuring better outcomes for children with OSA.
Expert opinion focusing on CV risk
Given the complexity of the disease and the vulnerability of the pediatric population, addressing OSA in children requires a holistic approach. We propose a strategy that goes beyond merely focusing on symptoms and traditional diagnostic methods. Instead, it should incorporate innovative tools that can provide insights into future CV risks and inform clinical decision-making. This approach to risk stratification can help reinforce treatment in some instances, avoid unnecessary interventions in children with mild-moderate OSA with low CV risk, or reassess the best alternative treatments for those whose risks or symptoms persist despite initial treatment. Figure 3 compares the standard of care with the proposal from an innovative CV perspective.
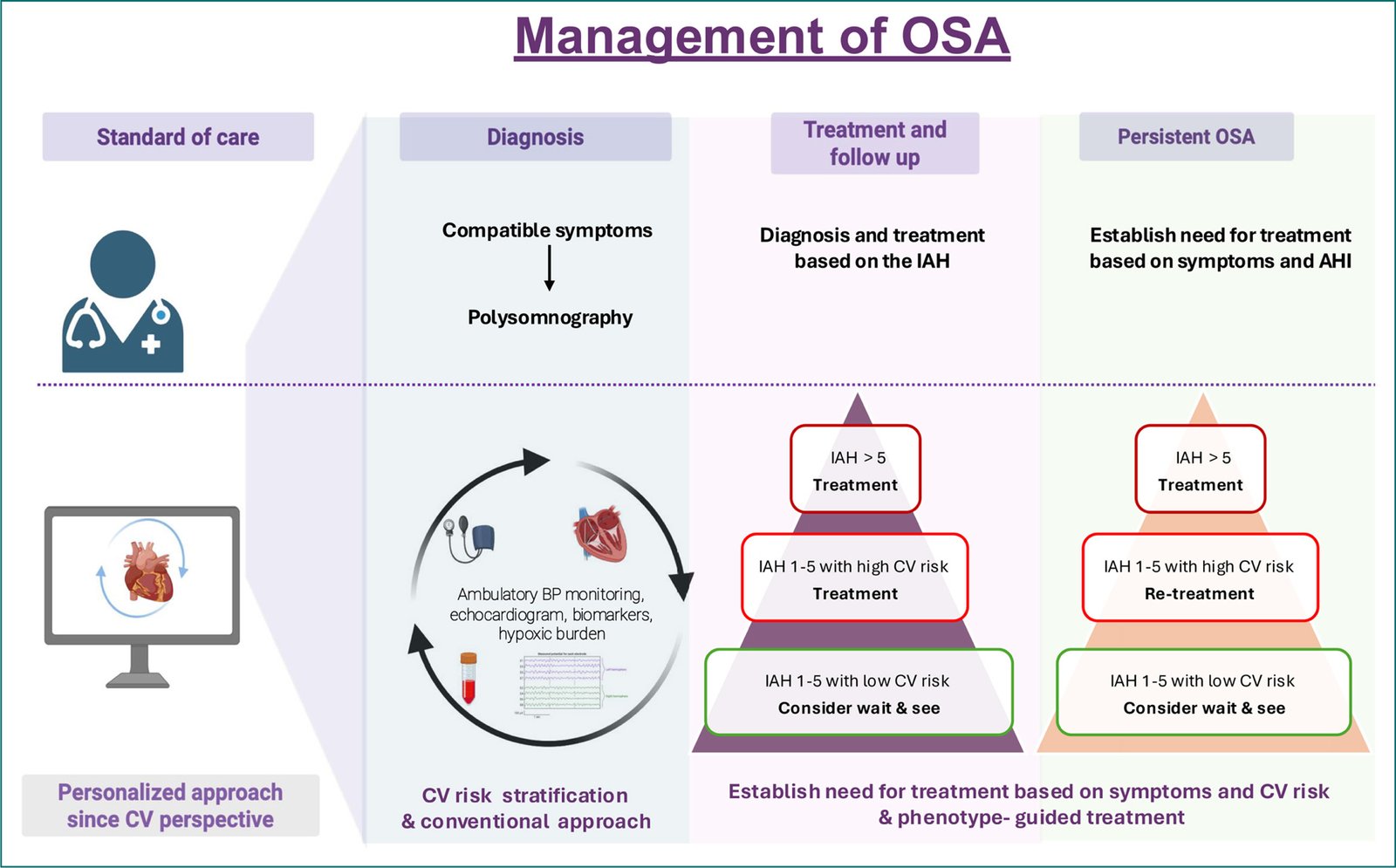
Figure 3. Algorithm for the management of pediatric OSA: current clinical practice versus a personalized approach since cardiovascular perspective. BP: blood pressure; CV: cardiovascular; IAH: index apnea hypopnea; OSA: obstructive sleep apnea; PSG: polysomnography.
All subjects with suspected OSA should be questioned about suggestive symptoms through a detailed history, as well as a thorough physical examination, to identify the underlying phenotype better and guide the best treatment.
We suggest that PSG be used as the diagnostic method, as guidelines recognize it as the gold standard. However, we understand the significant burden on healthcare resources and the critical need for early diagnosis and treatment in this vulnerable population. Therefore, in centers lacking adequately trained technical staff or resources to meet this demand, a simplified diagnostic method such as respiratory polygraphy may be considered to avoid delays in diagnosis. Although these simplified methods are not validated by all clinical practice guidelines, and the normal cutoff points can vary significantly among authors, they can serve as a screening tool to identify children with a high AHI who would clearly benefit from treatment. Children without an elevated AHI, whose diagnosis remains unclear, will require a PSG for clarification.
We have discussed the need to stratify children by future CV risk. Ideally, it would be beneficial to ensure that all children undergo ambulatory BP monitoring and incorporate HB into clinical practice. Ambulatory BP measurement has been robustly shown to be a good predictor of CV risk. It is safe and can help identify BP abnormalities and the presence of a NDP, which are key factors in determining treatment. It can be used after treatment to assess response and determine whether or not re-treatment or specific follow-up is needed.
Developing innovative parameters related to OSA and CV risk in the pediatric population could transform the management of children with OSA. These new measures may enhance our ability to predict which patients would benefit most from treatment. One promising approach is to assess HB as an alternative to the AHI for classifying the CV risk as a complementary tool. By correlating HB with BP values, we can identify specific groups of children who would derive the greatest benefit from OSA treatment, particularly in reducing future CV risks. Furthermore, it is an easily obtainable, reproducible parameter that can guide therapeutic decisions in children whose need for treatment is unclear and in whom CV risk stratification may be a determining factor. It is also helpful during follow-up to assess treatment response and support decision-making when OSA is persistent.
Along these lines, echocardiography can be a valuable tool for assessing CV risk and guiding diagnosis and follow-up. Although it is less accessible and its interpretation can be complex, the changes in cardiac function and structure observed in children with OSA present a new opportunity for exploration. In this sense, it can aid decision-making by helping predict which patients will benefit from treatment, based on baseline structural or functional alterations that are potentially reversible after treatment40–43. Echocardiography, combined with other biomarkers and traditional sleep studies, could help identify subjects who, in addition to an elevated AHI, have echocardiographic abnormalities that predict future CV risk and who could be monitored in the short and long term to assess treatment effectiveness.
As for serum and urinary markers of endothelial damage and inflammation, there are still aspects that need clarification. However, CRP, lipid profile, and microalbuminuria are inexpensive, accessible, and could be helpful in diagnosis and in monitoring to evaluate treatment response and help stratify CV risk at each stage.
Conclusion
OSA in children is a complex and heterogeneous disorder, and further research is needed to understand the underlying pathophysiological mechanisms responsible for its consequences. The pediatric population, being naïve to other CV diseases, represents an ideal target group for advancing knowledge of the CV risk associated with this condition. However, it is also a vulnerable population due to its ongoing development, making it crucial to establish unified diagnostic criteria and patient-centered therapeutic strategies. This personalized medicine perspective should rely on newly available tools, such as biosignals, such as HB, echocardiography, or biomarkers, although more studies are still required to validate their use in routine clinical practice. Their implementation is promising, as it could transform current clinical practice by helping to stratify both present and future CV risk in children with OSA and optimize treatment. Moreover, dealing with a pediatric population provides the opportunity to eliminate – or at least reduce – CV risk factors that could act as triggers for CV disease later in life. Although significant progress has been made in recent years, much remains to be learned, and further research in this field is clearly needed.
Acknowledgments
S. López-Monzoni: conceptualization, investigation, methodology, writing – original draft, review, and editing; L. Álvarez-Balado: conceptualization, methodology, writing – review, and editing; B. García-Mediano: conceptualization, methodology, writing – review and editing; O. Mediano: conceptualization, methodology, supervision, writing – review and editing.
Funding
This work was funded by Instituto de Salud Carlos III (ISCIII: PI18/00565 and PI22/01653) and co-funded by the European Regional Development Fund (ERDF)/“A way to make Europe”, the Spanish Respiratory Society (Sociedad Española de Neumología y Cirugía Torácica- SEPAR) (535-2018, 1073-2020, and 1425/2023), and an unconditional research grant from Menarini Laboratories and NEUMOMADRID (Sociedad Madrileña de Neumología y Cirugía Torácica).
Conflicts of interest
The authors declare no conflicts of interest.
Ethical disclosures
Protection of human subjects and animals. The authors declare that no experiments on humans or animals were performed for this research.
Confidentiality, informed consent, and ethical approval. This study does not involve personal patient data, medical records, or biological samples, and does not require ethical approval. SAGER guidelines do not apply.
Declaration on the use of artificial intelligence. During the preparation of this paper, the authors used Grammarly as an assistive AI tool to improve manuscript comprehension. After using this tool/service, the authors reviewed and edited the content as needed and take full responsibility for the publication’s content.